 
To determine the density of dry air at a given altitude we could use the relation 
Pressure is approximately halved for each additional increase of 56 km in altitude. And the higher you go into the atmosphere, the lower the pressure gets. This is because the density of air is proportional to the pressure and inversely proportional to temperature. But as altitude increases, the density drops dramatically. The density of dry air at sea level is 1.2929 kg/m 3 or about 1/800 the density of water. These are the freezing point (0 ☌) and normal air pressure at sea level (760 torr), respectively. Gases are usually compared at a standard temperature and standard pressure. The density of a substance (mainly gases) depends on temperature and pressure. Thus, gases have the smallest densities as compared to solids and liquids because gas molecules contain mostly empty space while molecules in liquids are more tightly packed together. Density can be obtained by dividing the mass (m) of an object by the volume of that object…Ī mass that is concentrated in a small volume has a greater density than a substance of equal mass that occupies a larger volume. "The density of air at sea level is approximately 1/800th the density of water."ĭensity (D) is the mass of a given volume of a substance. "The density of moist air may be determined by a similar relation: D = 1.2929 (273.13/T) where T is the absolute temperature B, the barometric pressure in mm, and e the vapor pressure of the moisture in the air in mm."ĬRC Handbook of Chemistry & Physics. "The density of dry air at sea level is about 1/800th the density of water."ĬRC Handbook of Chemistry & Physics. "Substance, Air, Mass Density (kg/m 3), 1.29, *Unless otherwise noted, densities are given at 0 ☌ and 1 atm pressure." The density has therefore increased by a factor of 1/.725, ie 1.Cutnell, John D. P = -K(dv/v) where K is the bulk modulus.įor a pressure, p = 0.1GPa, dv/v = -0.1 x 2.75 You can find the bulk modulus of various liquids in Kaye and Laby, Tables of Physical and Chemical Constants. To calculate this you will need the bulk modulus of the liquid. 
Liquids: For most purposes liquids are incompressible so density does not change with pressure but at very high pressures density will increase. Gases: If the fluid is a gas (and if the temperature remains constant), pressure is inversely proportional to volume for a given mass of gas, ie PV = C where C is a constant.Īs volume is the reciprocal of density, P/d = C where d is density. The higher the pressure the less the molecules will be able to move around thus it will be denser the lower the pressure the molecules can move around easier thus it will be less dense until it reaches the vapor point and turns to gas temperature also plays a factor the density of a fluid is inversely proportional to its pressure hence, as pressure is increased, a decrease in density is observed, mass etc. If the fluid is hot water, saturated, the density is higher by given higher pressure.įluid can either be gas or liquid. If the fluid is cold water, the same temperature, no change in density. If the fluid is saturated steam, the density (kg/m3) is higher by given higher pressure while the specific steam volume (m3/kg) is lower. 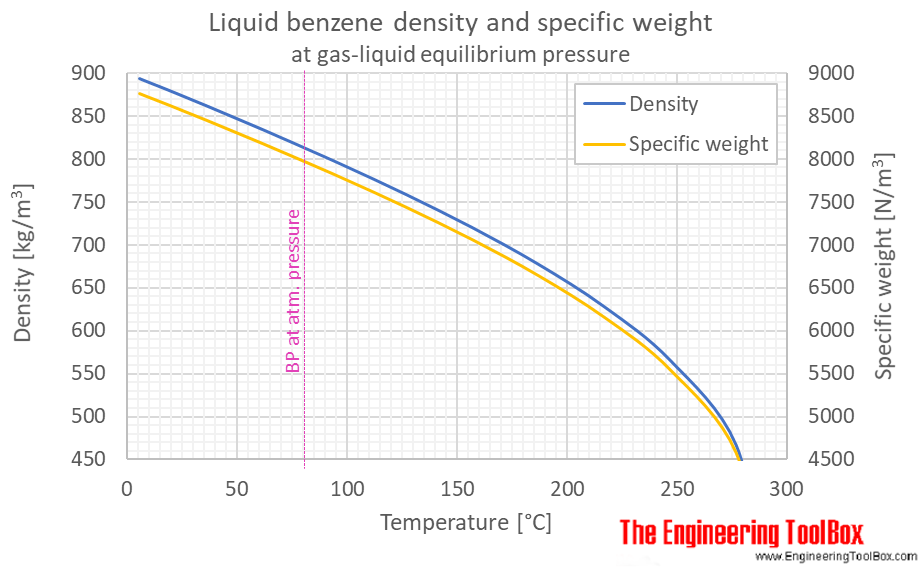
Pressure and density are directly proportional (all other factors constant) for a gas.įluid is a constant under any pressure, unlike gas 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
